Where the quantum number is the subshell, the magnetic number m represents the number of possible values for available energy levels of that subshell as shown in the table below. L z, the component of angular momentum in the z direction, is given by the formula:Īnother way of stating the formula for the magnetic quantum number ( m l =, …, 0, …, ) is the eigenvalue, J z=m lh/2π. The quantum number m refers to the projection of the angular momentum for any given direction, conventionally called the z direction. To show that only certain discrete amounts of angular momentum are allowed, has to be an integer. To show each of the quantum numbers in the quantum state, the formulae for each quantum number include Planck's reduced constant which only allows particular or discrete or quantized energy levels. 
This causes the wave to display particle-like packets of energy called quanta. The energy of any wave is the frequency multiplied by Planck's constant. Where is Planck's reduced constant, also called Dirac's constant. To describe the magnetic quantum number m you begin with an atomic electron's angular momentum, L, which is related to its quantum number by the following equation: It was first demonstrated by two German physicists, Otto Stern and Walther Gerlach. This phenomenon is known as space quantization. More precisely, for a given orbital momentum quantum number (representing the azimuthal quantum number associated with angular momentum), there are integral magnetic quantum numbers m ranging from to, which restrict the fraction of the total angular momentum along the quantization axis so that they are limited to the values m. Given a particular, m is entitled to be any integer from up to. The magnetic quantum number m does not affect the electron's energy, but it does affect the probability cloud. The quantum number m refers, loosely, to the direction of the angular momentum vector. The magnetic quantum number associated with the quantum state is designated as m. 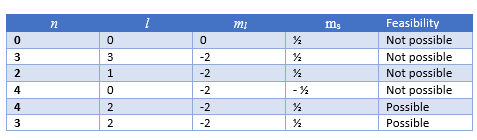
The magnetic quantum number arose in the solution of the azimuthal part of the wave equation as shown below. Therefore, the equations for the first three quantum numbers are all interrelated. The wavefunction of the Schrödinger wave equation reduces to the three equations that when solved lead to the first three quantum numbers. The four quantum numbers n, l, m, and s specify the complete and unique quantum state of a single electron in an atom called its wavefunction or orbital. There are a set of quantum numbers associated with the energy states of the atom. Essential Laboratory Skills Guide Derivation
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
